A New Dawn for Spinal Cord Injury Recovery
Spinal cord injuries (SCIs) are life-altering, often leaving individuals with permanent loss of movement, sensation, or both. For decades, treatment options were limited to physical therapy and symptom management, offering little hope for reversing damage. Enter stem cell therapy—a groundbreaking frontier in medical science that promises to rewrite the narrative for SCI patients. By harnessing the body’s own regenerative potential, this innovative approach is sparking optimism, backed by compelling research and clinical trials. Let’s explore how stem cell therapy is transforming the landscape of spinal cord injury recovery with hope, science, and resilience.
The Science Behind Stem Cells
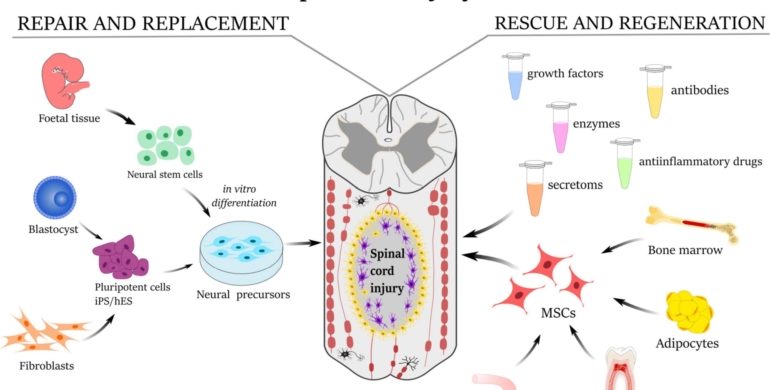
Stem cells are the body’s master builders, capable of transforming into specialized cells like neurons, muscle, or bone. Their unique ability to self-renew and differentiate makes them ideal candidates for repairing damaged tissues, including the intricate neural networks of the spinal cord. In SCIs, the cord’s delicate nerve fibers are often severed or crushed, disrupting communication between the brain and body. Unlike other tissues, the spinal cord has limited natural repair capacity, making stem cell therapy a game-changer. Mesenchymal stem cells (MSCs), embryonic stem cells (ESCs), and induced pluripotent stem cells (iPSCs) are among the most studied for SCI. Each type offers distinct advantages, from MSCs’ anti-inflammatory properties to iPSCs’ patient-specific compatibility, reducing rejection risks. Research from institutions like the Mayo Clinic shows that stem cells can promote nerve regeneration, reduce inflammation, and create a supportive environment for healing.
The Promise of Regeneration
Imagine a therapy that could rebuild the very pathways that enable movement and sensation. Stem cell therapy aims to do just that by replacing damaged neurons, supporting existing ones, and fostering tissue repair. A 2023 study published in Nature Neuroscience found that transplanting neural stem cells into animal models with SCIs led to significant motor function recovery within weeks. The cells integrated into the injury site, forming new neural connections and reducing scar tissue, a major barrier to recovery. Clinical trials, such as those conducted by Asterias Biotherapeutics, have shown similar promise in humans. In one phase I/II trial, patients with complete cervical SCIs who received oligodendrocyte progenitor cells derived from ESCs regained measurable motor function in their upper limbs within six months. These findings suggest that stem cells could bridge the gap where traditional treatments fall short, offering a lifeline to those with severe injuries.
Overcoming the Challenges
While the potential is immense, stem cell therapy for SCIs is not without hurdles. The spinal cord’s complex environment, with its mix of inflammation, scar tissue, and limited blood supply, poses significant challenges. Timing is critical—administering stem cells too soon after injury may exacerbate inflammation, while waiting too long risks irreversible damage. A 2024 meta-analysis in The Lancet highlighted that optimal outcomes occur when therapy is initiated within the first two weeks post-injury. Another hurdle is ensuring the right cell type and delivery method. Intrathecal injections, direct transplantation, or biomaterial scaffolds are being explored, each with trade-offs. For instance, scaffolds can guide cell growth but may introduce foreign materials, risking immune responses. Ethical concerns also linger, particularly with ESCs, though iPSCs—derived from a patient’s own cells—are alleviating some of these issues. Despite these challenges, ongoing research is refining techniques, making therapy safer and more effective.
Real-World Impact and Patient Stories
The true measure of stem cell therapy’s potential lies in the lives it touches. Consider the story of Kris Boesen, a young man who, in 2016, suffered a traumatic cervical SCI that left him paralyzed from the neck down. As part of a clinical trial at the University of Southern California, Kris received an injection of 10 million AST-OPC1 stem cells. Within months, he regained the ability to move his arms, feed himself, and even use his phone—a monumental leap for someone facing quadriplegia. While not all patients experience such dramatic recoveries, stories like Kris’s underscore the therapy’s transformative potential. A 2025 report from the Christopher & Dana Reeve Foundation noted that over 1,500 SCI patients worldwide have participated in stem cell trials, with 60% showing some functional improvement. These numbers, while preliminary, highlight a growing body of evidence that stem cells can restore what was once thought lost.
The Role of Technology and Innovation
Advancements in technology are propelling stem cell therapy forward. CRISPR gene editing, for example, is being used to enhance stem cells’ regenerative capabilities by targeting specific genes involved in neural repair. A 2024 study from Stanford University demonstrated that CRISPR-edited iPSCs improved axonal regeneration in mice by 30% compared to unedited cells. Biomaterials, such as hydrogels, are also revolutionizing delivery methods by creating a supportive “scaffold” for stem cells to thrive in the injury site. Additionally, 3D bioprinting is emerging as a tool to create precise tissue constructs that mimic the spinal cord’s architecture, potentially improving cell integration. These innovations are not just theoretical—companies like Neuralstem are integrating them into clinical pipelines, with phase II trials showing promising results in chronic SCI patients. The synergy of biology and technology is paving the way for more effective, personalized treatments.
The Road Ahead
The future of stem cell therapy for SCIs is bright but requires careful navigation. Regulatory bodies like the FDA are scrutinizing therapies to ensure safety and efficacy, a process that can delay widespread access. As of 2025, only a handful of stem cell treatments for SCI have reached phase III trials, with full approval still years away. Cost is another barrier—therapies can range from $50,000 to $200,000 per treatment, often not covered by insurance. Yet, global investment in regenerative medicine is surging, with over $2 billion allocated to SCI research in 2024 alone. Collaborations between universities, biotech firms, and patient advocacy groups are accelerating progress, ensuring that trials are more inclusive and diverse. The hope is that within the next decade, stem cell therapy will become a standard option, not just an experimental one, for SCI patients worldwide.
A Call for Hope and Action
Stem cell therapy for spinal cord injuries represents more than a medical breakthrough—it’s a beacon of hope for millions. From restoring movement to improving quality of life, the potential is undeniable, grounded in rigorous science and real-world results. While challenges remain, the pace of innovation is relentless, driven by researchers, patients, and advocates united in a common goal. For those living with SCI, the message is clear: the future is not about accepting limitations but about redefining possibilities. As research advances and therapies become more accessible, we stand on the cusp of a new era in SCI recovery—one where the impossible may become reality. Visit our website https://stemnovanetwork.com/ to Buy Stem Cell Therapy for Spinal Cord Injury at Affordable Price.